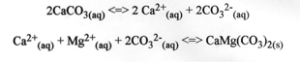Frequently Asked Questions
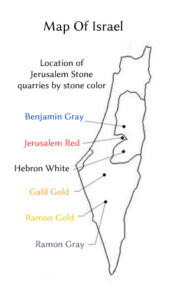
What is Jerusalem Stone? Jerusalem Stone is a term describing natural stone coming from inside the area of the Jordan River to the Mediterranean Sea, and from the Red Sea to the Galilee.
How much absorption by weight, density, and flexural strength? Technical Information ASTM, Ramon Gold, Absorption by Weight% C97 value=0.78, Density in lbs./cu. ft C97 value=169 Flexural Strength psi C880-M value=1,248, Compressive Strength psi C170 value=11,490, Abrasion Resistance Hardness C241 value=10.5.
Natural Stone of Israel